U.S. Pharma Tariffs And MFN Become Law After April 2 Update
By Mathini Ilancheran, senior delivery lead - research, R&D, Beroe Inc.

On April 2, 2026, President Trump signed a sweeping executive order converting over a year of tariff threats into binding trade law. Issued under Section 232 of the Trade Expansion Act of 1962, the proclamation imposes a 100% ad valorem tariff on patented pharmaceutical products and their active pharmaceutical ingredients (APIs), effective July 31, 2026 for large companies and September 29, 2026 for smaller manufacturers.1,2 Generic pharmaceuticals, biosimilars, and certain specialty products, including orphan drugs, remain exempt for now, with a formal review of generics mandated within one year.2
This is not a price shock. It is a structural reset. For sponsors, CROs, CDMOs, and central labs, the clock is running.
The Policy Architecture
The proclamation establishes a tiered tariff framework. The rates are determined by country of origin, whether the importing company has a Commerce-approved onshoring plan, and whether it has a signed MFN pricing agreement with HHS. [2]
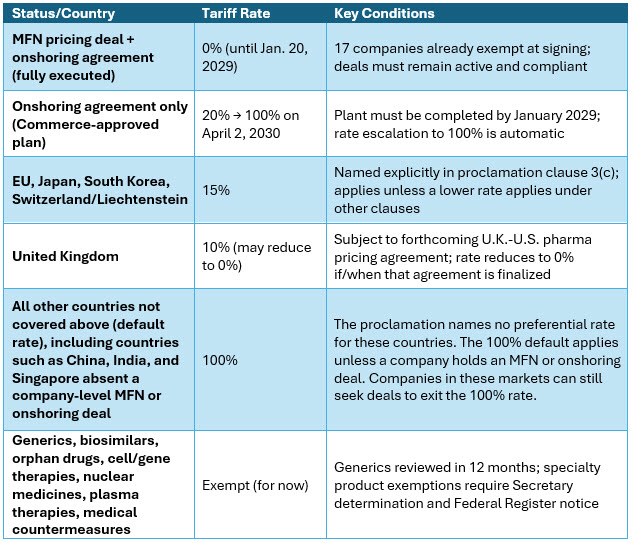
Source: White House Presidential Proclamation, April 2, 2026, clauses 3(a)–3(e) and clause 11.2 Note: The proclamation does not explicitly name China, India, or Singapore — these countries fall under the default 100% rate by not being listed in any preferential tier.
Three structural features distinguish this order from the October 2025 tariff framework.10
- Tariff exposure is now primarily a function of company-level MFN deal status, not geography alone; a fully executed agreement confers a 0% rate regardless of manufacturing origin.2
- The 20% onshoring rate is transitional, not stable; it escalates automatically to 100% on April 2, 2030.2
- The White House reports that Section 232 pressure has already triggered approximately $400 billion in new domestic pharma investment commitments, which will rapidly compete with the U.S. manufacturing capacity that sponsors are simultaneously trying to reserve.1
Impact Across The Drug Development Value Chain
The value chain exposure mapped in the October 2025 framework 10 remains directionally valid. The April 2 order introduces additional nuance at each stage, particularly where MFN deal status now intersects with country of origin risk.
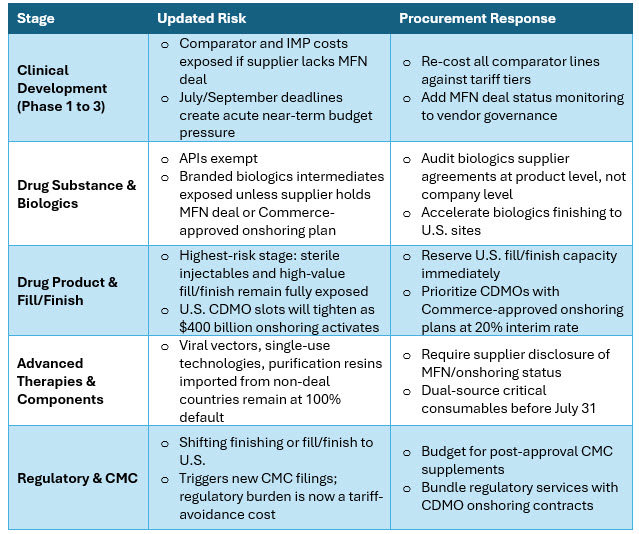
Derived from White House Proclamation2; ITIF analysis7; updated from prior framework10,11
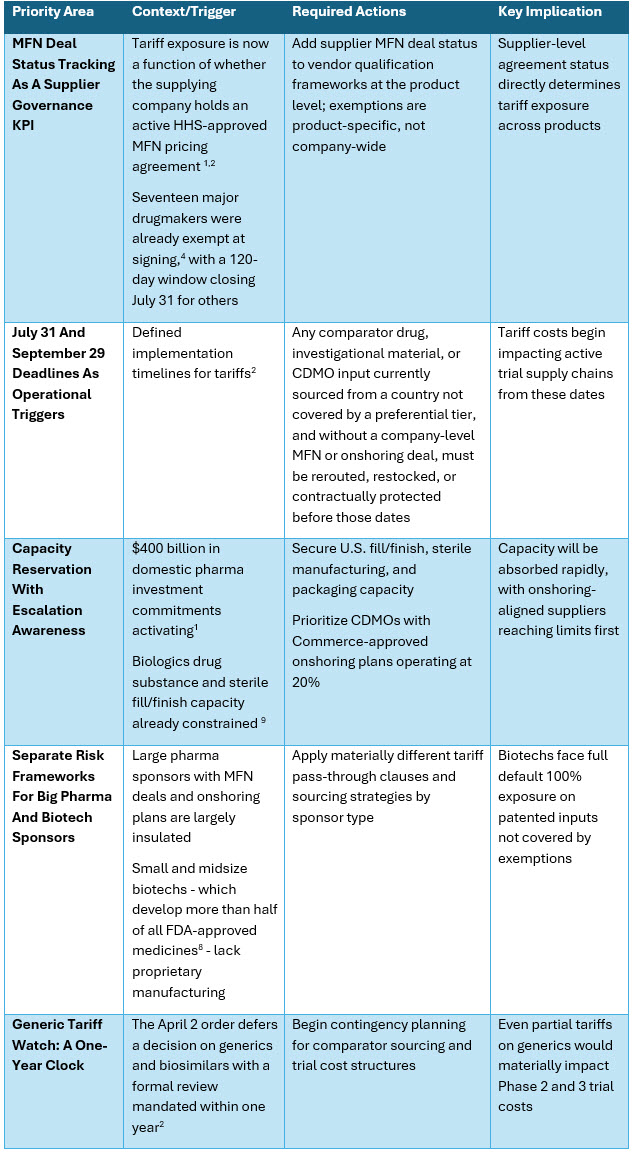
Strategic Procurement Priorities
Five priorities require immediate action, building on the eight-point framework in the October 2025 analysis.10
Conclusion
The April 2, 2026 executive order converts 12 months of Section 232 tariff pressure into enforceable trade law. The default 100% rate applies to all countries not explicitly listed in the proclamation's preferential tiers, and the proclamation names only the EU, Japan, South Korea, Switzerland/Liechtenstein, and the U.K. All other manufacturing origins fall under the default rate unless a company-level MFN or onshoring deal applies.2
For sponsors, CROs, CDMOs, and the full outsourced network, the immediately actionable priorities are:
- Map MFN deal status across all supplier relationships at the product level.
- Re-cost trial budgets against the July 31 and September 29 effective dates, and reserve U.S. fill/finish capacity before $400 billion in onshoring commitments compete for the same slots. 1,2
The organizations that treat these deadlines as operational triggers and not compliance exercises will turn tariff volatility into a sourcing and capacity advantage.
References:
- White House, "Fact Sheet: President Donald J. Trump Bolsters National Security and Strengthens U.S. Supply Chains by Imposing Tariffs on Patented Pharmaceutical Products," April 2, 2026. Available: https://www.whitehouse.gov/fact-sheets/2026/04/fact-sheet-president-donald-j-trump-bolsters-national-security-and-strengthens-u-s-supply-chains-by-imposing-tariffs-on-patented-pharmaceutical-products/
- White House, "Adjusting Imports of Pharmaceuticals and Pharmaceutical Ingredients into the United States," Presidential Proclamation, April 2, 2026. Available: https://www.whitehouse.gov/presidential-actions/2026/04/adjusting-imports-of-pharmaceuticals-and-pharmaceutical-ingredients-into-the-united-states/
- CNBC, "Trump administration sets up to 100% tariffs on some imported drugs, with many companies exempt," April 2, 2026. Available: https://www.cnbc.com/2026/04/02/trump-pharmaceutical-tariffs-100percent.html
- Axios, "Trump hits drugmakers with tariffs on some pharmaceutical drugs," April 2, 2026. Available: https://www.axios.com/2026/04/02/trump-drug-prices-tariffs
- The Hill, "Trump signs order imposing 100 percent tariff on brand name drugs," April 2, 2026. Available: https://thehill.com/policy/healthcare/5813970-trump-signs-drug-tariffs/
- Pharmaceutical Executive, "President Trump Imposes 100% Tariffs on Branded Pharmaceuticals," April 3, 2026. Available: https://www.pharmexec.com/view/president-trump-imposes-100-tariffs-branded
- ITIF, "Trump Pharma Tariffs: Wrong Rx for U.S. Patients, Manufacturing, and Innovation," April 3, 2026. Available: https://itif.org/publications/2026/04/03/trump-pharma-tariffs-wrong-rx-for-us-patients-manufacturing-and-innovation/
- BIO, "BIO Statement on the Section 232 Pharmaceutical Proclamation," April 2026. Available: https://www.bio.org/press-release/bio-statement-section-232-pharmaceutical-proclamation
- PharmaSource, "What to Expect in Pharma Manufacturing in 2026," January 2026. Available: https://pharmasource.global/content/expert-insight/what-to-expect-in-pharma-manufacturing-in-2026-industry-leaders-share-their-predictions/
- M. Ilancheran, "100% U.S. Drug Tariffs: Implications for Pharma and Outsourcing," Clinical Leader / Beroe Inc., Oct. 17, 2025. Available: https://www.clinicalleader.com/doc/u-s-drug-tariffs-implications-for-pharma-and-outsourcing-0001
- M. Ilancheran, "U.S. Pharma Tariffs and MFN in 2026: Manufacturing and Procurement Impact," Clinical Leader / Beroe Inc., Feb. 18, 2026. Available: https://www.clinicalleader.com/doc/u-s-pharma-tariffs-and-mfn-in-manufacturing-and-procurement-impact-0001
About The Author:
 Mathini Ilancheran is a Research Manager and an expert in market intelligence and industry analysis, specializing in delivering strategic insights that help Fortune 500 companies make informed decisions. With a focus on global pharma, biotech, and medical devices, she brings deep expertise in value chain analysis, industry and technology trends, competitive intelligence, and strategy. Mathini has authored 45+ publications on R&D outsourcing, offering actionable perspectives that guide global enterprises in optimizing outsourcing practices, category management, and long-term planning.
Mathini Ilancheran is a Research Manager and an expert in market intelligence and industry analysis, specializing in delivering strategic insights that help Fortune 500 companies make informed decisions. With a focus on global pharma, biotech, and medical devices, she brings deep expertise in value chain analysis, industry and technology trends, competitive intelligence, and strategy. Mathini has authored 45+ publications on R&D outsourcing, offering actionable perspectives that guide global enterprises in optimizing outsourcing practices, category management, and long-term planning.
