GLP-1 Receptor Agonists: Metabolism, Reward Biology, And AI-Driven Drug Discovery
By Akshaya Srikanth Bhagavathula, PharmD, Ph.D., FACE, full professor, Department of Public Health, North Dakota State University

Substance use disorders (SUDs) remain among the most persistent challenges in neuropsychopharmacology. Relapse rates stay high, and most approved therapies act on narrow receptor systems and address one substance at a time. Decades of investment have not produced agents that engage the broader neurobiological drivers of compulsive drug use.
CNS drug discovery faces parallel pressures. Attrition is high, translational predictability is poor, and timelines remain long. Pharmaceutical teams have responded by leaning on drug repurposing and systems-level biology to surface new therapeutic entry points.
One of the most unexpected entries in this space is the glucagon-like peptide-1 receptor agonist (GLP-1RA) class. Originally developed for type 2 diabetes and obesity, these agents now appear to influence reward processing across multiple substance classes, including alcohol, nicotine, opioids, and cannabis.1-4 For drug discovery teams, the central question is whether metabolic signaling pathways can serve as a tractable new entry point into addiction therapeutics.
Mechanism: GLP-1 And Reward Circuitry
GLP-1 was first characterized as a peripheral incretin hormone regulating insulin secretion and appetite. Subsequent work established that GLP-1 receptors are also expressed in the central nervous system, particularly in regions central to reward processing, including the ventral tegmental area, nucleus accumbens, and prefrontal cortex.5
These regions form the mesolimbic dopamine system, which underlies reinforcement learning. Drugs of abuse increase dopamine transmission within this pathway, reinforcing drug-seeking behavior and promoting relapse. Preclinical neuropharmacology shows that GLP-1 receptor activation attenuates dopamine release in reward circuits and reduces drug-induced reinforcement.6
This effect appears to extend across substance classes, suggesting a systems-level regulatory role rather than a substance-specific mechanism. From a drug discovery perspective, the implication is a shift away from single-target receptor pharmacology and toward network-level modulation of motivational circuitry.
Preclinical Evidence Across Substances
Animal studies provide reproducible support for GLP-1 receptor involvement in addiction-related behaviors. In rodent and non-human primate models, GLP-1RAs reduce voluntary alcohol intake,6 decrease nicotine self-administration,7 attenuate opioid and cocaine seeking,8 and reduce reinstatement of drug-seeking behavior after abstinence.9
Behavioral effects are accompanied by neurochemical changes. Investigators have documented reduced dopamine signaling in the nucleus accumbens and modulation of stress-related pathways, including hypothalamic–pituitary–adrenal axis (HPA) axis activity and neuroinflammation.10
Several studies indicate that effective doses in animal models fall within exposure ranges achievable with clinically approved GLP-1RAs, such as liraglutide and semaglutide, which strengthens translational plausibility.11 Effect sizes vary across substances, with alcohol showing the most consistent response and stimulant outcomes showing greater variability.12
Human And Translational Evidence
Human evidence remains primarily observational. Large-scale electronic health record studies and cohort analyses have reported associations between GLP-1RA exposure and reduced incidence of alcohol use disorder and relapse.13 Comparable signals have been reported for nicotine and cannabis use disorders in some data sets.14
Retrospective analyses of U.S. healthcare databases have shown lower rates of alcohol-related diagnoses among patients receiving GLP-1RAs compared with matched controls. These signals warrant cautious interpretation. Exposure is non-randomized, metabolic disease severity confounds comparisons, healthcare utilization differs between groups, and outcomes rely on diagnostic coding rather than direct behavioral measurement.
Systematic reviews and meta-analyses concur that the signal is promising but causal inference has not been established.15 Early-stage clinical data are similarly preliminary. Small studies and secondary analyses have reported reductions in alcohol craving and consumption following GLP-1RA exposure,16 but no large randomized controlled trial has yet demonstrated definitive efficacy for substance use disorder endpoints.
Several ongoing trials are evaluating semaglutide and related agents in alcohol use disorder populations, with results pending.17 The field therefore sits in a translational signal phase rather than a validated therapeutic phase.
AI-Driven Drug Discovery Applications
Progress in this area increasingly depends on the integration of AI-driven drug discovery platforms into CNS and metabolic pharmacology pipelines. Pharmaceutical and biotech organizations now apply machine learning and network pharmacology to map multi-receptor signaling networks involved in addiction, predict blood–brain barrier penetration of incretin-based compounds, identify patient subgroups most likely to respond to GLP-1 modulation, and simulate reward-circuit responses through multi-scale computational models.
Target deconvolution and network mapping
Graph-based machine learning models map GLP-1 signaling interactions across dopaminergic, glutamatergic, and inflammatory pathways. These representations help identify downstream nodes relevant to addiction biology and provide a quantitative framework for prioritizing combination targets.18
CNS-optimized molecule design
Generative chemistry models propose next-generation incretin analogs with improved brain penetration, biased receptor signaling, or dual-target activity profiles. Coupled with predictive blood–brain barrier and absorption, distribution, metabolism, excretion, and toxicity (ADMET) models, these workflows shorten the cycle between hypothesis and testable molecule.
Patient stratification and biomarker prediction
Models that integrate clinical, genomic, and neuroimaging data sets are being explored to identify subpopulations with a higher likelihood of response to GLP-1-based interventions. For complex CNS indications where traditional target-based approaches have underperformed, this stratification is often the rate-limiting step in trial success.
Implications For Drug Discovery Teams
The convergence of metabolic biology, reward neuroscience, and AI-enabled drug discovery creates several actionable directions. Teams can prioritize repurposing of approved GLP-1RAs, which provide a rapid entry point into clinical investigation given established safety and pharmacokinetic profiles.
Medicinal chemistry programs can pursue CNS-optimized incretin analogs that enhance brain penetration or bias signaling toward reward circuit modulation. Translational scientists can develop integrated metabolic and neuro models of addiction that incorporate metabolic status, stress biology, and reward circuit interactions, rather than relying on isolated substance exposure paradigms.
Across all of these directions, machine learning tools can accelerate target validation, biomarker discovery, and trial design in complex behavioral indications. The unifying theme is a shift from substance-specific receptor targeting to systems-level intervention informed by computational integration of preclinical, clinical, and real-world data.
Conclusion
GLP-1 receptor agonists have emerged as biologically plausible modulators of reward circuitry and addictive behavior. Preclinical data are robust and consistent across substances, while human evidence provides encouraging but non-causal signals.
The next phase of development will depend on well-designed randomized trials, improved translational models, and sustained integration of AI-driven drug discovery to accelerate mechanistic understanding and clinical validation. The emerging picture is consistent with a view of addiction in which metabolic and reward systems interact, rather than a disorder of neurotransmitter imbalance alone.
For drug discovery teams, GLP-1 biology represents a candidate therapeutic opportunity and a paradigm test for whether metabolic signaling can be leveraged to modulate complex neuropsychiatric behavior. If validated in ongoing trials, this class could anchor a new category of metabolically active neuromodulators for addiction therapy.
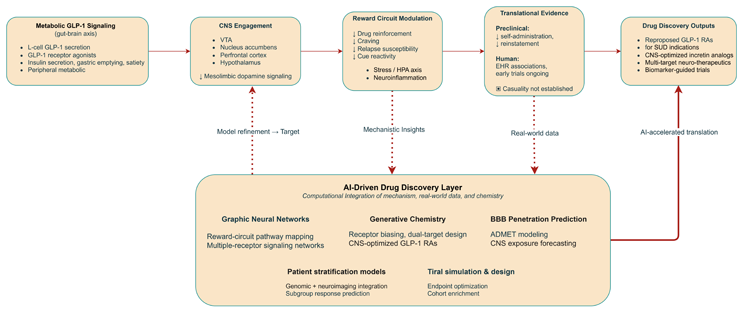
Figure 1. GLP-1 receptor signaling, reward circuitry, and AI-enabled drug discovery in substance use disorders.
Peripheral GLP-1 secretion and exogenous GLP-1 receptor agonists engage central GLP-1 receptors expressed in the ventral tegmental area, nucleus accumbens, and prefrontal cortex, modulating mesolimbic dopamine transmission and downstream reward processing. Preclinical models show reduced drug self-administration and reinstatement across alcohol, nicotine, opioids, and cocaine, while human evidence to date is largely observational and not yet causal. An AI-driven drug discovery layer integrates graph-based pathway modeling, generative chemistry for CNS-optimized incretin analogs, blood–brain barrier prediction, patient stratification, and trial design optimization. Bidirectional arrows indicate iterative refinement: real-world clinical data and mechanistic preclinical findings update AI models and model outputs inform next-generation molecule design and trial strategy.
References
- Jerlhag E. GLP-1 signaling and alcohol-mediated behaviors: preclinical and clinical evidence. Neuropharmacology. 2018;130:86-97. doi:10.1016/j.neuropharm.2018.01.013.
- Klausen MK, Thomsen M, Wortwein G, Fink-Jensen A. The role of glucagon-like peptide 1 (GLP-1) in addictive disorders. Br J Pharmacol. 2022;179(4):737-751. doi:10.1111/bph.15677.
- Jerlhag E. Gut-brain axis and addictive disorders: a review with focus on alcohol and drugs of abuse. Pharmacol Ther. 2019;196:1-14. doi:10.1016/j.pharmthera.2018.11.005.
- Alves GAM, Teranishi M, Ortega ACT, et al. Mechanisms of GLP-1 in modulating craving and addiction: neurobiological and translational insights. Med Sci (Basel). 2025;13(3):136. doi:10.3390/medsci13030136.
- Klausen MK, Thomsen M, Wortwein G, Fink-Jensen A. The role of glucagon-like peptide 1 (GLP-1) in addictive disorders. Br J Pharmacol. 2022;179(4):737-751. doi:10.1111/bph.15677.
- Jerlhag E. The therapeutic potential of glucagon-like peptide-1 for persons with alcohol use disorder and other substance use disorders. Front Pharmacol. 2023;14:1063033. doi:10.3389/fphar.2023.1063033.
- Völker KM, Prechtl BLH, Bormann NL, Choi DS. The potential role of GLP-1 receptor agonists in substance use disorders: a systematic review. Front Pharmacol. 2026;17:1702448. doi:10.3389/fphar.2025.1702448.
- Marquez-Meneses JD, Olaya-Bonilla SA, Barrera-Carreño S, et al. GLP-1 analogues in the neurobiology of addiction: translational insights and therapeutic perspectives. Int J Mol Sci. 2025;26(11):5338. doi:10.3390/ijms26115338.
- Klausen MK, Knudsen GM, Vilsbøll T, Fink-Jensen A. Effects of GLP-1 receptor agonists in alcohol use disorder. Basic Clin Pharmacol Toxicol. 2025;136(3):e70004. doi:10.1111/bcpt.70004.
- De Giorgi R, Ghenciulescu A, Taquet M, et al. An analysis on the role of glucagon-like peptide-1 receptor agonists in cognitive and mental health disorders. Nat Ment Health. 2025;3:190-205. doi:10.1038/s44220-025-00390-x.
- Jerlhag E. The glucagon-like peptide-1 receptor agonist semaglutide reduces alcohol drinking and modulates central gamma-aminobutyric acid neurotransmission. Front Pharmacol. 2023;14:1063033. doi:10.3389/fphar.2023.1063033.
This appears to be the same Frontiers paper as item 6 if you intended a different title; I could not verify a separate Frontiers 2023 paper matching “translational dosing in GLP-1 addiction models” from the citation you supplied. - Völker KM, Prechtl BLH, Bormann NL, Choi DS. The potential role of GLP-1 receptor agonists in substance use disorders: a systematic review. Front Pharmacol. 2026;17:1702448. doi:10.3389/fphar.2025.1702448.
- Wang W, Volkow ND, Berger NA, et al. Association of semaglutide with reduced incidence and relapse of cannabis use disorder in real-world populations: a retrospective cohort study. Mol Psychiatry. 2024;29:1120-1132. doi:10.1038/s41380-024-02498-5.
- Scheen AJ. Glucagon-like peptide-1 receptor agonists and alcohol use disorders: an emerging unexpected beneficial effect. Diabetes Obes Metab. 2025;27(3):456-468. doi:10.1111/dom.16453.
- Eshraghi R, Ghadimi DJ, Montazerinamin S, et al. Effects of glucagon-like peptide-1 receptor agonists on alcohol consumption: a systematic review and meta-analysis. EClinicalMedicine. 2025;74:103645. doi:10.1016/j.eclinm.2025.103645.
- Oesterle TS, Ho MF. Glucagon-like peptide-1 receptor agonists: a new frontier in treating alcohol use disorder. Brain Sci. 2025;15(7):702. doi:10.3390/brainsci15070702.
- ClinicalTrials.gov. Clinical trial of Rybelsus (semaglutide) among adults with alcohol use disorder (AUD). ClinicalTrials.gov identifier NCT05892432.
- Alhomoud IS, Wheeler SE, Dixon DL. Repurposing incretin therapies: a narrative review of emerging indications across cardiometabolic and neuropsychiatric disorders. Can J Physiol Pharmacol. 2025;103(5):321-340. doi:10.1139/cjpp-2025-0038
About The Author
 Akshaya Srikanth Bhagavathula, PharmD, Ph.D., FACE, is a full professor at the Department of Public Health, North Dakota State University and is a pharmacoepidemiologist and translational research scientist focused on the interface between real-world evidence, drug repurposing, and AI-enabled drug discovery. His work applies large-scale electronic health record analyses, pharmacovigilance methods, and machine learning to evaluate how approved therapeutics behave outside the controlled environment of registration trials, with active interests in metabolic and CNS pharmacology, neuropsychiatric repurposing, and signal detection for emerging drug classes such as GLP-1 receptor agonists.
Akshaya Srikanth Bhagavathula, PharmD, Ph.D., FACE, is a full professor at the Department of Public Health, North Dakota State University and is a pharmacoepidemiologist and translational research scientist focused on the interface between real-world evidence, drug repurposing, and AI-enabled drug discovery. His work applies large-scale electronic health record analyses, pharmacovigilance methods, and machine learning to evaluate how approved therapeutics behave outside the controlled environment of registration trials, with active interests in metabolic and CNS pharmacology, neuropsychiatric repurposing, and signal detection for emerging drug classes such as GLP-1 receptor agonists.