Crystallization Process Development: Ramping Up Bench-Scale Control For Manufacturing
By Hayley Reece, Executive Director, Technical Services, and Craig Callahan, PhD, Project Scientist 4, Crystallization Process Development, Cambrex

Designing a crystallization process to control drug substance and drug product quality.
Targeted solvent selection. Selecting the right solvent is critical to ensure optimum yield, polymorphic form, impurity purging and effective isolation and drying.
Controlled crystallization: Transferring methods from the bench to the plant
Crystallization is an essential element of drug development, whether it is performed during formation of an API or as a means to purify intermediates. Since physical structure and biological activity are directly linked, controlling the physical form of a compound is paramount to a final drug product’s quality and effectiveness. Drug manufacturers must also demonstrate to regulatory authorities that the physical properties and chemical purity of a drug substance are well understood and controlled. Therefore, the development of a robust crystallization process is an integral part of drug development.
Before scientists can manipulate various critical process parameters to yield the optimal result, they must have an in-depth understanding of the thermodynamic and kinetic processes affecting the product material. At Cambrex, we leverage our expertise to bridge the gap between the bench and manufacturing, providing controlled crystallization processes to help our clients achieve clinical delivery timelines.
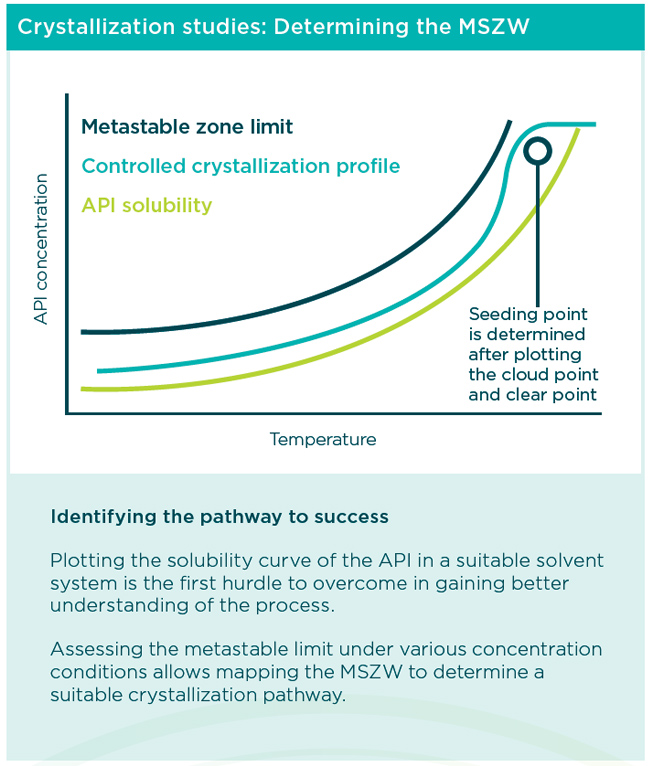
Identifying critical operating conditions: Solvent selection
The emphasis on quality of starting material has dual advantages by contributing to meeting regulatory requirements and helping to ensure successful scale-up for manufacturing. Obtaining a pure crystal form of an API is exacting work, and it requires advanced expertise to first identify the optimal solid form and then consistently isolate the stable product. Our experts achieve the optimal form and crystal habit by controlling several operating conditions that impact crystal formation, including evaluating the solvent system, determining the metastable zone width (MSZW) of the process, and controlling nucleation and crystal growth.
Initial characterization is performed when receiving crude starting material or material from an existing process to establish a benchmark for comparison. Using standard analytical techniques, data is collected to characterize crystallinity and polymorphic form, morphology, presence of solvents, water content, chemical integrity, and purity.
Solvent selection is the first critical step in developing a process for crystallization. Scientists evaluate solubility data across a wide screen of solvents and solvent mixtures based on multiple factors including material recovery, favorable polymorphic form and other particle properties, as well as the ability to purge impurities. Regulatory requirements surrounding the safety profile of solvents are also considered when choosing the most suitable systems to advance in the process development
In some cases, single crystal x-ray diffraction (SXRD) provides increased understanding of the crystal structure, which is used to narrow the range of suitable solvents for further study based on expected hydrogen bond interactions. A solvent addition method is performed first to approximate solubility, and to further target the list of solvents that merit more detailed screening experiments.
Mid-scale testing: Initial crystallization to controlled reactors
With a targeted list of potential solvent systems in hand, scientists can begin small-scale trials to further understand the crystallization process. Comparison studies allow an initial assessment of cooling and anti-solvent methods to assess if and how they affect yield, purity and particle morphology. Many additional process parameters including hold periods, initial concentrations, and seeding protocol are monitored during small scale crystallizations (300 to 500mg) to allow optimization prior to scale up.
Once the scientists have confidence in the fundamental process, the process development is advanced to a controlled reactor. Our experts are industry leaders in vessel design and utilize engineering experience to execute modeling studies and carefully match vessel geometries to the plant models. During their evaluation, they consider diameter ratios of the vessel and the impeller as well as other reactor vessel components like impeller type and baffle location. By gathering precise specifications of the manufacturing equipment, they can use sophisticated software to model a matching smaller-scale reactor during development. This increases the transferability of the process when scaling for manufacturing.
During controlled reactor experiments, measurements are taken to plot the MSZW to better understand nucleation and dissolution during cooling and heating, and to determine a suitable seeding protocol. Using cooling and heating profiles, the solubility curve and metastable zone limit are plotted to define the zone between clear points and cloud points. Determining the MSZW gives scientists an indication of how best to control the crystallization process, and it is critical to gain sufficient process understanding. By defining the operational limits of crystal nucleation, scientists can then optimize other steps in the process within this window.
Following MSZW assessments, the critical crystallization process parameters are determined with the use of online process analytical tools. This allows monitoring of the effects of each parameter on process control, and how they ultimately affect the properties of the isolated product material. In addition, a drying protocol is developed with an assessment of the impact of drying operations on product quality.
Demonstrate early control. Drug producers can’t expect to control their process in the plant without first controlling the process in the lab.
Spotlight: Moving the needle on particle morphology
The ultimate goal of developing a crystallization process in drug development is to transfer methods for successful manufacture. At its core, technology transfer is the transfer of knowledge to replicate the process and produce consistent results. For crystallization development, knowledge of the physical properties and behavior of the crystal must be considered alongside the vessel geometries and other reactor components to devise a successful process.
In this case, the morphology of the crude starting material immediately steered upstream efforts for improvement of downstream processing. Polarized Light Microscopy (PLM) revealed a thin, needle-like crystal morphology, and the team recognized the negative impact this would have on both isolation and further handling and formulation processes. Initial efforts focused on developing a crystallization method that would yield a more desirable crystal morphology.
Screening was performed to understand the approximate solubility and determine a primary list of 24 potentially suitable solvents and anti-solvents. With this in hand, our experts began crystallization trials evaluating cooling and anti-solvent addition methods. This narrowed the list to 7 potential solvent systems that yielded more uniform crystal size and improved morphology. The studies also revealed that slow cooling was optimal to control the rate of nucleation and allow production of larger, lower aspect ratio crystals. Secondary solvent screens determined the optimal solvent systems and ratios for further investigation.

Next, specific solvents were assessed for thermodynamic solubility in 2-point screens, allowing the team to focus further studies on 6 solvent systems in specific ratios at specific temperatures. Analysis of these slurries included solid form characterization and HPLC measurement of concentration and purity, all of which contributed to selecting the best system and steering further optimization. After this round of screening, a targeted list of 3 ICH Class 3 solvents exhibited promising suitability based on solubility, recovery, and consistency in morphology. These systems were then evaluated against an expanded range of temperatures to determine a more detailed solubility curve.
Using the most promising solvent systems, the metastable zone widths were determined allowing suitable seeding points and protocols to be established. While one of the solvent systems exhibited a narrow zone that would require strict controls during crystallization and increase risk of loss of process control, another showed a wider zone allowing for a more lenient process and greater operating window. Fine-tuning other parameters to optimize the crystallization included assessment of cooling profiles, anti-solvent addition rates, volumes and temperatures, hold periods, seeding protocol and mixing dynamics. Each of these parameters was evaluated using continuous online monitoring (PAT) for their effects on product quality to achieve the optimal crystal habit, yield, polymorphic form and product purity. During this stage of observation, our experts also monitored the rate of filtration, washing protocols and the effect of different drying methods.
The team also replicated the reactor vessels and each component that would be used during manufacturing to ensure successful technology transfer. Our experts gather thorough specifications and requirements of the equipment in the plant to recreate the small-scale reactors used during development. In the end, our team delivered a firmly controlled process to begin manufacturing for clinical demand.
Beyond the crystal ball: Delivering a blueprint for success
Crystallization is no longer merely a separation method. Experts now have a greater understanding of the individual factors that contribute to optimal crystal formation and the tools to monitor several process parameters in-situ. While crystallization efforts are traditionally associated with small molecules, Cambrex also maintains expertise in peptide crystallization, adding to our command in this space of drug development.
Control over crystallization signifies control over the factors that impact the solid form, and it follows that achieving control of nucleation and crystal growth can determine success or failure of scale-up crystallizations. Furthermore, alignment with QbD principles helps maximize efficiency while minimizing risk to the integrity of the final product. This case study highlights the value of leveraging expertise to bring bench-scale chemistry to the manufacturing plant. By replicating the results achieved in customized small reactors, our clients have the tools to implement an optimized and well-controlled crystallization process that stands up to scaling the process for manufacturing demand. In addition, it positions drug producers to meet increasing regulatory demand to demonstrate control over the processes that can impact the quality and safety of drug substances.
About Cambrex
Cambrex is a leading global contract development and manufacturing organization (CDMO) that provides comprehensive analytical and IND enabling services, as well as drug substance development and manufacturing across the entire drug lifecycle.
With over 40 years of experience and a team of 2,000 experts servicing global clients from North America and Europe, Cambrex is a trusted partner in branded and generic markets for API development and manufacturing. Cambrex offers a range of specialized drug substance technologies and capabilities, including continuous flow, controlled substances, solid-state science, material characterization, and highly potent APIs.
