ARIAD Corrects Diabetes in Mice Through Two-Pronged Delivery Technique
ARIAD's new technology, called "RAPID" (Regulated Accumulation of Proteins for Immediate Delivery), is based on a proprietary method of storing pre-made therapeutic proteins inside cells as inactive aggregates or clusters. Stored proteins are apparently stable in this form inside the cell, but they can be released through action of an oral, small-molecule drug that breaks the protein clusters apart. Because the proteins are pre-made in the cell, they are released almost immediately in fully active form in response to administration of ARIAD's small-molecule drug. The level of protein secretion is directly dependent on the dose of the drug; the larger the dose, the greater the secretion. Without the drug, the protein is secreted at a pre-established basal level, which in some applications is essentially zero.
In the Science paper human insulin was engineered to accumulate in specific cells in diabetic mice. These mice cannot make their own insulin and consequently have elevated blood sugar levels. Within fifteen minutes of administering ARIAD's oral drug, insulin appeared in the bloodstream and rapidly rose to peak levels within less than two hours, leading to a correction of blood sugar. The level of insulin then subsided over the next two hours and returned towards basal levels.
This experimental study demonstrates that it may be possible to replace repeated daily injections of recombinant insulin with a one-time or infrequent delivery of a vector containing a modified insulin gene, followed by sublingual or oral administration of an ARIAD small-molecule drug at mealtimes and as needed in response to circulating blood sugar levels.
RAPID complements ARIAD's other proprietary technology for controlling gene expression, ARGENT (ARIAD Regulated Gene Expression Technology). With ARGENT, circulating levels of protein, such as erythropoietin and human growth hormone, peak after about 24 hours and then are sustained within a therapeutic window for a prolonged period of time. ARIAD scientists have previously demonstrated long-term production of erythropoietin and increased numbers of red blood cells (needed to treat anemia) for more than 450 days in rhesus monkeys using ARGENT and adeno-associated viral (AAV) vectors. RAPID products also can be delivered by AAV vectors, which contain no viral genes and are non-pathogenic.
"Along with ARGENT, RAPID enhances our broad portfolio of innovative technologies for regulated delivery of therapeutic proteins," said Harvey Berger, Chairman and CEO of AREAD. "RAPID further strengthens ARIAD's position in regulated gene therapies for multiple diseases and provides several new product opportunities for the company.
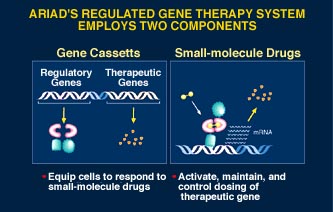
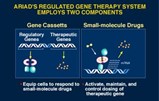
Turning Gene Therapy On and Off
The major problem facing gene therapy is how to turn it on and off. For example diabetics only require therapeutic insulin when they eat carbohydrates. Controlling the expression of genetically programmed proteins through a drug taken orally gives control to the patient or physician. Specific benefits include:
- Consistent and therapeutically relevant levels of gene expression.
- Controlled dosing of therapeutic proteins, leading to high therapeutic efficacy and low systemic toxicity.
- Safety—Regulated gene therapy may allow for tight control over the production of potent proteins with systemic toxicity. As an extra measure of safety, engineered cells can be eliminated from the body through programmed cell death.
- Allows gene therapy to be administered as a conventional pharmaceutical—Since the gene therapy product is controlled through an orally available drug, therapy may be controlled by physicians in the same way as other pharmaceuticals.
- Easily definable products with ongoing markets—Drugs used to control therapeutic protein production will be required by each patient for as long as the patient undergoes gene therapy.
For more information: Jay R. LaMarche, Chief Financial Officer, Ariad Pharmaceuticals Inc., 26 Landsdowne St., Cambridge, MA 02139. Tel: 617-494-0400, ext. 202.
Angelo DePalma
