Key Concepts Driving The Future Of Single-Use In Biopharmaceutical Production
By Matthew Olsen
Single-use systems consisting of bags, tubing, filters and other connecting components have now been in use in the biopharmaceutical industry for over 25 years. There are nearly no areas of the process where at least one single-use solution does not exist to provide the many conveniences which disposable, pre-sterilized components can enable. The quick turnaround times, reduced capital costs and significantly reduced validation costs associated with single-use technologies have now been fully embraced by the industry and are well illustrated by the speed of adoption these technologies have enjoyed. New entrants to the industry are now able to benefit from a “Single-Use First” business plan and avoid the costly buildout of a fully stainless steel reusable plant. The disruptive advantages this confers are analogous to the advantages in telecom buildout speed and cost that have been achieved in the developing world through the use of mobile phone technology to completely leap-frog the need to build out expensive wired telecommunications infrastructure. The adoption of single-use in combination with the prevalence of Contract Manufacturing Organizations (CMO) can result in a significant reduction in the timelines and up-front investment required to bring a novel therapeutic to the clinic and the market.
Although their adoption has been swift and unit operations which were formerly groundbreaking are becoming commonplace, the familiarity and experience of end users with the available products is in turn leading to a grassroots demand for new advancements in the science and technology of single-use bioprocessing. These demands have been expressed, for instance, by the BioPhorum Operations Group (BPOG) community which has published a number of documents representing a set of explicit requests and roadmaps developed by end users for advancements in biopharmaceutical production, some of which provide feedback on single-use technologies.
This article will discuss four topics related to single-use bioprocessing which are commonly cited by end users as amenable to future development. These concepts are key to the future of single-use systems in the midst of the current paradigm shift to the 100% single-use bioprocess:
Single-use systems consisting of bags, tubing, filters and other connecting components have now been in use in the biopharmaceutical industry for over 25 years. There are nearly no areas of the process where at least one single-use solution does not exist to provide the many conveniences which disposable, pre-sterilized components can enable. The quick turnaround times, reduced capital costs and significantly reduced validation costs associated with single-use technologies have now been fully embraced by the industry and are well illustrated by the speed of adoption these technologies have enjoyed. New entrants to the industry are now able to benefit from a “Single-Use First” business plan and avoid the costly buildout of a fully stainless steel reusable plant. The disruptive advantages this confers are analogous to the advantages in telecom buildout speed and cost that have been achieved in the developing world through the use of mobile phone technology to completely leap-frog the need to build out expensive wired telecommunications infrastructure. The adoption of single-use in combination with the prevalence of Contract Manufacturing Organizations (CMO) can result in a significant reduction in the timelines and up-front investment required to bring a novel therapeutic to the clinic and the market.
Although their adoption has been swift and unit operations which were formerly groundbreaking are becoming commonplace, the familiarity and experience of end users with the available products is in turn leading to a grassroots demand for new advancements in the science and technology of single-use bioprocessing. These demands have been expressed, for instance, by the BioPhorum Operations Group (BPOG) community which has published a number of documents representing a set of explicit requests and roadmaps developed by end users for advancements in biopharmaceutical production, some of which provide feedback on single-use technologies.
This article will discuss four topics related to single-use bioprocessing which are commonly cited by end users as amenable to future development. These concepts are key to the future of single-use systems in the midst of the current paradigm shift to the 100% single-use bioprocess:
- Increased Understanding of Biocompatibility
- Improved Integrity Assurance for Single-Use Systems
- Integration of Single-Use Measurement and Automation to support PAT efforts
- Development of a Resilient Global Supply Network

Figure 1: A completely single-use cell culture process train is possible with bioreactor volumes up to the 2000L scale.
Increased Understanding of Biocompatibility
Biocompatibility of single-use systems can refer to the assurance of compatibility with cell culture as well as compatibility with any downstream product streams and process conditions. In cell culture applications, any single use components must allow cell culture to proceed nominally whereas for downstream applications biocompatibility concerns tend towards ensuring protein stability and minimizing leachables. Any plastic components being offered for use in single-use bioprocessing equipment are assumed to be USP 87 & 88 compliant, produced in compliance with EMA 410/01 and available with a full set of extractables data. USP 87 and 88 are in-vitro and in-vivo biological reactivity tests to show that a given part meets USP Class VI standards and compliance with EMA 410/01 indicates that the plastics present are processed to minimize the likelihood of transmission of prions capable of causing spongiform encephalopathy diseases. These estimations of biocompatibility provide a clear picture to evaluate compatibility and have become the minimum requirements for consideration for use. Since all products offered to the market now meet this requirement, the bar has been raised on what can be considered an enhanced approach to biocompatibility. As we move to a condition in which all vendors are providing extractables data gathered and presented in a similar format, areas that are still available for enhancement are the tools to help the evaluator of single-use technologies to determine the leachables profile of a specific single-use assembly design as well as increased understanding and control of particulates in single-use assemblies including visible particulates.
As mentioned earlier, the requests of end users voiced by the BPOG initiative have convinced most single-use assembly providers to tailor their extractables data to the format specified in the BPOG extractables guide. Harmonization of extractables datasets improves interpretation and comparison of the extractables produced by plastic films under multiple sets of conditions but the datasets don’t take into account an entire assembly and thus only represent one aspect of evaluation of leachables from a full single-use assembly. One potential enhancement for ease of evaluation by the end user would be the provision of a comprehensive extractables guide for an individual assembly to ease evaluation of potential leachables from the entire single-use product in the manufacturing process. This sort of BOM-specific extractables profile represents the next evolution of extractables data provision.
Once one has the extractables data package for the bag film in hand, an important consideration during evaluation of the data is how representative this data is in the context of ongoing film production. How many and what sorts of controls are in place to ensure that the one-time data provided in an extractables package is representative of product from ongoing production at time points potentially years away from the original extractables assessment? Specifications for all raw materials and control of the production of both the plastic resin as well as the bag film can be key to ensuring that any data provided will remain valid in the future.
Specification of the formulation of plastic resins and films ensures that any extractables encountered will be well characterized since all components of the formulations are known beforehand. When the formulation of the plastic resins and additives are known in detail, the extractables studies can better target, quantify and identify the extractable profiles of the film under study. While this may seem obvious, gaining a view into the full formulation of all plastics being used in production of single-use bags is not always possible and thus it is worthwhile to emphasize the advantages when possible.
Once all raw materials are fully understood, control of the production processes for the film then ensures that any extractables present will be consistent between lots of product. One approach to ensuring a reliable operational space is identified for all critical processing parameters is the use of a design of experiments type approach to ensure the design space is fully understood. The operational parameters and their effects can therefore be fully understood. This understanding ensures that any validation studies and toxicological assessments remain valid and reproducible from lot-to-lot. The control of the resins and all other critical process steps such as extrusion, welding and gamma radiation ensure lot-to-lot consistency of extractable profiles. The extractable data contained in these guides thus remain representative and valid along the product life cycle.
Particulates are another area with room for improvement to allow for ease of adoption of single-use systems in critical applications. The often stated goal for particulates in single-use systems is to minimize particulates as much as possible while recognizing it is not possible to achieve complete removal of all particles from any product. Historically, qualification data on particulates in single-use systems has been provided using an adapted USP 788 protocol which provides a framework for evaluation of sub-visible particulate load but does not address visible particulates. As such, the only stated goal for visible particulates was their reduction along with complete removal if observed. At this point, some guidelines have been published by industry associations but no standards exist for visible particulates in single-use systems although some related standards have been proposed.1,2
While visible particles will be removed by any downstream 0.2 micron filtration steps they are still of concern in protein production processes and minimization of their presence is always preferable. As single-use continues its expansion into all areas of the bioprocess including fill/finish applications, the assurance of reduction of visible particulates has become an area of focus as some single-use parts used in final filling might contact product after the final 0.2 micron filtration. New or expanding product categories which cannot perform final sterilizing grade filtration, such as cell therapies, also have increased requirements for reduced particulate load.
No matter what the current inherent particulate load of a single-use system, manufacturers can always examine their current situation and improve it over time through the adoption a continuous improvement workflow. A systematic study of background particulate load accompanied by a detailed source identification procedure can yield a tremendous amount of actionable information and process insight. Once the sources of particulates are understood, procedures for reduction can then be undertaken. Once new strategies are in place, the particulate load can then be assessed again and the procedure can be begun again. The BPSA has released a document which provides recommendations for manufacturers of single-use systems as well as end users for testing, evaluation and control of particulates.3
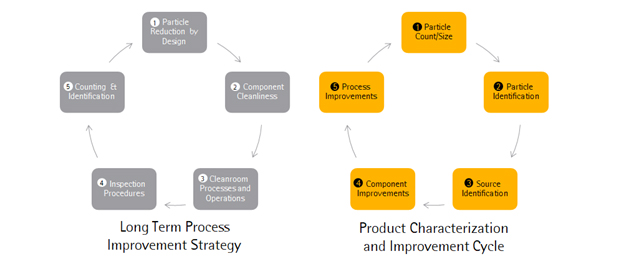
Figure 2: Continuous Improvement in Particulate Counts requires a combination of Long Term Strategy for Process Improvement combined with constant Monitoring and Identification of Particulate Sources.
In addition to the reduction of sources of particulate contamination over time, there is also room for innovation in the area of detection of particulates within an assembly during manufacture. Visual inspection on light tables remains the industry standard for visible particulate detection so improvements in this area can provide greater assurance that any visible particulates present will be detected and either removed or cause the diversion of the assembly.
There can be no way to guarantee the absence of particulates within a given assembly but the adoption of a continuous improvement approach to particle reduction is a worthy extension of current efforts and will yield improvements if conducted appropriately. The assessment and continued reduction in particulate load of all consumables is a key area of development for single-use system suppliers with an intent to provide products for the most critical applications.
Improved Integrity Assurance for Single-Use Systems
Reliable assurance of Container Closure Integrity (CCI) has become an area of increased interest from end users as well as regulatory agencies as single-use bags continue to advance into commercial cGMP production and are used in increasingly critical steps of the process. Simple inspection of single-use assemblies by eye can detect gross defects that will cause a liquid leak but by definition visual inspection cannot catch all defects that might cause a liquid leak or contamination. To improve this situation, new strategies for container closure integrity based upon pressure decay tests using air or other gas tracers have been developed. Recent guidelines and norms have been published or are being finalized for ensuring increased safety via integrity assurance and testing methods.4,5,6,7,8 This interest has led to increased end user requests for greater development in the area of risk mitigation to allow for usage of single-use products in critical applications.
An initial area of interest to improve integrity assurance is with the use of Pre-use Point-of-Use Leak tests although they can only form part of an overall Container Closure Integrity Assurance Strategy. Air leak tests performed on site are able to detect gross defects and those as small as 10 – 50 microns in the tested parts of the assembly depending upon the size of the bag in question using an assembly similar to that shown in Figure 3 below. This is sufficient to detect gross defects where a film is punctured by an outside item which are the sort most likely to occur during shipping and handling. Punctures of the film from handling are typically 200 microns or larger. Bag damage during shipping and handling and the loss of production material have been revealed as the primary concerns of the industry by several end-users surveys on single-use and thus point of use leak tests can be useful for providing improved end user confidence.9,10 A leak test at the point of use provides assurance that there will be no large defects present which might cause liquid leaks during subsequent processing of critical high value products. However, point-of-use leak tests alone are not sufficient to provide complete assurance of integrity.
One strategy to increase final assembly integrity assurance beyond that of a single point of use air leak test is to also perform a very sensitive integrity test during manufacture to ensure all possible manufacturing defects have been detected prior to shipment to the end user. Helium Leak Testing performed on the final assembly at the site of manufacture is one such method that can reliably detect much smaller defects down to as little as 2 microns. It is important to perform a risk assessment for each type of potential application but 2 microns has been demonstrated as the maximum allowable leakage limit for single use systems under a number of applications and therefore this test ensures that all tested bags are integral as they leave the factory.
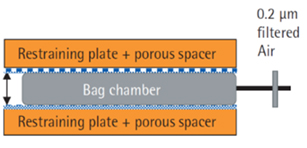
Figure 3: Bag Leak Testing equipment based upon recommendations of ASTM F-2095.
A combination strategy employing a very sensitive gas tracer test during manufacture together with a pressure decay air leak test on site provides a reliable measure of single-use assembly integrity and represents the most advanced CCI strategy possible for risk mitigation. The gas tracer testing ensures integrity prior to shipment from the site of manufacture and the point of use test can detect any possible defects which could occur by subsequent handling as their size will always be detectable by those on-site tests.
Integration of Single-Use Measurement and Automation to support PAT efforts
As biopharmaceutical manufacturers come to rely upon single-use equipment in all unit operations, the need for robust and scalable in-line and in-situ process sensors that are compatible with that equipment becomes increasingly critical. In the past, some compromise often had to be made as compared to reusable technologies when attempting to design a completely single-use unit operation such as ultrafiltration but the increased availability of single-use sensors is removing any need to compromise for certain scale processes. Single-use assembly manufacturers are responding to the requests of their end users and are creating a variety of approaches to in-line process analytical technologies. Due to continued development in sensor technology, there are now single-use sensors available that can provide reliable monitoring of nearly all unit operations used in typical bioprocesses. The technology has developed to such an extent that in some applications there now exists a second generation of single-use sensors which provide improved performance over their first generation single-use counterparts. Many of these sensors are available in both flow-through and in-bag measurement formats although some styles remain proprietary to only one supplier. Table 1 below provides a list of sensors available in a completely single-use format suitable for biomanufacturing as of this writing.
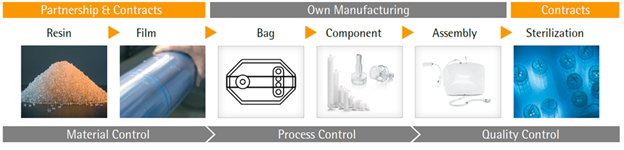
Table 1: Single-Use Sensors are used for different purposes throughout the process
The need for sensors in single-use bioreactors drove much of the initial development in this area but now these same sensors or adapted versions of them are moving their way downstream. When the first single-use bioreactors were introduced to the industry, reusable sensors were incorporated into the consumables due to the dearth of available single-use sensors. As time has gone on and the selection of single-use bioreactors has increased, completely single use solutions are now more commonly available for cell culture. In addition, many downstream unit operations are now supported with standard single-use product offerings.
The availability of in-line and in-situ sensors for each of these parameters has led to increased demand for standard consumables with integrated sensors and equipment platforms designed for inclusion of these sensors. A number of manufacturers have developed platforms allowing for local control of the unit operation as well as incorporation of single-use unit operations into the larger data acquisition and process control system. These standard consumables and platforms are driving the implementation of PAT in single-use systems.
Development of a Resilient Global Supply Network
The first three concepts discussed above represent extensions of the technical knowledge or product base for the industry but there are additional non-technical developments that are required for the advancement of the single-use industry. The continued expansion of the global footprint of the biopharmaceutical industry necessitates the further development of the single-use supply chain network for seamless production of single-use items around the world. End users are building manufacturing networks for biopharmaceutical products that span the globe and they want to ensure they can produce equivalent product in a reasonable timeframe from all of these facilities when tech transfers are taking place. In a single-use manufacturing environment, single-use systems form the backbone of the manufacturing operations and it therefore becomes critical that equivalent single-use assemblies can be received at each site since as these become the primary process contact materials. At the same time, consolidation on the vendor side has resulted in a smaller quantity of suppliers of single-use assemblies and components and those suppliers have likewise worked to broaden their footprints to match the increasingly global biopharmaceutical production.
In addition to the global presence of demand for pharmaceutical grade single-use systems, an additional driver for development of global supply networks is the increasingly global distribution of biopharma process development and production. Global tech transfers both to and from CMO’s are a critical area that all single-use suppliers must be prepared to support. Biopharmaceutical manufacturers have increasingly located their R&D and Process Development operations in widely distributed biotech hotbeds while locating manufacturing where needed around the globe. It is critical that all suppliers be able to support these process development personnel with the assurance that any single-use products they evaluate be available with minimal leadtime around the globe. One can always ship items around the globe from a single location but it makes increasing sense to establish a global supply network to ensure the consistent supply of high quality single-use products around the world in a timely and efficient manner.
A supply network can become more resilient through an increase in the number of nodes as well as the implementation of risk management strategies on the raw materials. The primary risks to the network result from disruption to production in a single manufacturing site or delays or disruptions to the network of suppliers that all single-use manufacturers rely upon. Both of these eventualities require the development of specific strategies to ensure any disruptions are short-lived and minimal in scale.
Five strategies that can be considered for development of a resilient global supply network are detailed below. These methods can ensure both robust final product manufacturing capability as well as assurance of raw material supply for single-use bag systems.
- Establishment of Resin Specifications and Process Controls
- Establishment of Film Extrusion Design Space and Process Controls
- Long Term Supply Contracts
- Multiple Manufacturing Sites
- Full Manufacturing Process Controls from Resin to final Products
Responsible manufacturers are using long term supply contracts and quality agreements with critical suppliers to ensure supply continuity for their raw materials and components. An agreement with the supplier of a plastic resin can be difficult for single-use suppliers to put in place but is the gold standard for assurance of quality resin supply. The next step in the supply of single use items is the production of the plastic components from the resin materials. In order to ensure that single-use products can be produced with consistent mechanical properties and other quality attributes, it is important to investigate the possible production space using the principles of design of experiments. Through investigation of the system design space one can ensure that materials will be produced with consistent mechanical parameters as well as consistent secondary characteristics such as extractables. One additional ancillary advantage of operating within a validated design space is that it allows for a streamlined expansion of the supply network to additional manufacturing sites. The development of this design space approach represents a step forward in the production of single-use technologies and provides much greater assurance of quality supply to end users around the globe.
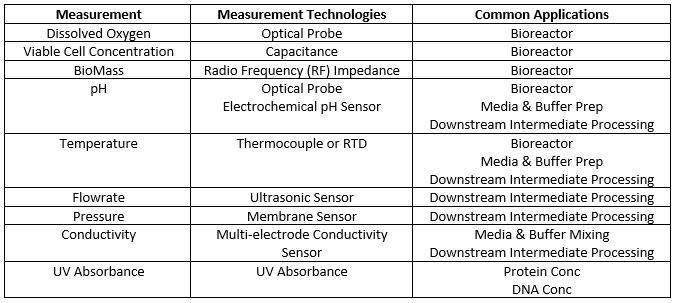
Figure 4: Suppliers must better characterize raw materials & improve controls along the entire supply chain in order to improve the assurance of quality supply for single-use systems.
There are advantages inherent in the structure of a network that cannot be replicated by a single-site manufacturing plan. The availability of a distributed manufacturing network enables the fast transfer of production between manufacturing sites for capacity balancing as well as business continuity assurance in the case of supply disruption from a single site. A single source manufacturing site will always possess a greater risk profile regarding disruption by a site-specific emergency. Manufacturers of Single-Use systems continue to strengthen and expand their manufacturing networks and capabilities as they have realized that a reliable supply base is a strategic advantage when all end users are seeking reliable global suppliers.
The continued build out of individual suppliers’ single-use supply networks offers numerous advantages for end users in the form of risk mitigation, quality assurance and global supply assurance. Suppliers that are not able to ensure their products will be available whenever needed around the world are at a disadvantage as biopharmaceutical production becomes an increasingly global endeavor.
Summary
The four developing areas of single-use science and technology highlighted here are critical to supporting the accelerating transition to a fully single-use biopharm production environment. Developments in Biocompatibility and Integrity Assurance allow for increased use of trusted single-use systems in high criticality areas of the bioprocess such as fill-finish operations. The increasing availability of sensor solutions for single-use applications allows for seamless integration of single-use into the production environment while meeting traditional expectations for real-time process monitoring and control. And lastly, the development of a truly global supply network for single-use solutions allows biopharmaceutical manufacturers to develop their processes where their expertise lies with the expectation that the tech transfer of their process to any site globally can be seamlessly executed by the single-use vendor community. Continued advancement in these highlighted areas of the science and technology of single-use will ensure that the single-use processing paradigm can remain relevant for new product niches within the biopharma space such as cell and gene therapies and antibody drug conjugates.
Endnotes
- American Society for Testing and Materials, “Standard Practice for the Extraction of Particulate Contamination from Single Use Components,” Work Item 54630, ASTM, West Conshohocken, PA.
- Parenteral Drug Association, “Visible Particle Measurement for Aseptic Single-Use Bags for Rubber Components,” PDA Harmonization Effort, PDA, Bethesda, MD.
- Bio-Process Systems Alliance, “Recommendations for Testing, Evaluation and Control of Particulates from Single-Use Process Equipment,” BPSA, Arlington, VA (2014)
- United States Pharmacopoeia, “Proposed revision to general chapter sterile product packaging integrity evaluation,” USP <1207>, USP, Rockville, MD, (September 2014 publication)
- American Society for Testing and Materials, “Standard practice for Integrity Assurance & Testing of Single-use Systems,” ASTM E55 WK64337, ASTM, West Conshohocken, PA.
- American Society for Testing and Materials, “Test Method for Microbial Ingress Testing on Single-use Systems,” ASTM E55 WK64975, ASTM, West Conshohocken, PA.
- Bio-Process Systems Alliance, “Design, Control, and Monitoring of SUS for Integrity Assurance,” BPSA, Arlington, VA (July, 2017).
- American Society for Testing and Materials, ASTM F2095-07 Standard Test Methods for Pressure Decay Leak Test for Flexible Packages With and Without Restraining Plates.
- BioPlan Associates, Inc, “12th Annual Report and Survey of Biopharmaceutical Manufacturing Capacity and Production”, Rockville, MD, (April 2015).
- Aspen Brook Consulting, LLC, “Aspen Brook 6th Annual Survey of the single use Bioprocessing Market 2014,” Aspen Brook Consulting, LLC, Park City, UT, (2014).
©Originally published in Chemical Engineering Process
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Drug Discovery Online? Subscribe today.
