Peptide Synthesis: Delivering Cost-Effective Strategies
By Brian Heasley, PhD, Director, Process Chemistry, Cambrex

Meeting the rising demand for peptide therapeutics with liquid phase peptide synthesis.
Tip the scale. Cambrex chooses strategies that allow you to benefit from economies of scale in procurement of raw materials.
Clinical potential of peptides: The role of CDMOs in a growing pipeline?
As early as the 1920s, when isolated insulin was first used as a replacement therapy, peptides have been acknowledged as a valuable class of pharmaceutical compounds for their ability to promote natural therapeutic pathways. Despite their desirable profile combining high selectivity with low toxicity, technological advances in peptide synthesis have only recently caught up to provide manufacturing methods to meet demand. Now, CDMOs can draw upon multiple synthesis strategies to offer large-scale production of peptide APIs across a wide range of therapeutic applications.
A notable example demonstrating the promise of large-scale production of peptides was the launch of Fuzeon, a large 36-mer peptide involved in HIV-1 fusion inhibition, which was produced on commercial scale by a process involving solid phase peptide synthesis (SPPS). Pricing of Fuzeon may have hindered its success in the market, which is an inherent issue with SPPS: it is difficult to scale up without excessive cost. The requirements for specialized equipment, excess reagents and starting materials, additional purification steps, and other factors, all add up to increased cost of production.
As drug candidates progress through development and clinical stages, the required batch volumes grow, which is a critical factor driving API synthesis strategy. For peptide APIs, using a liquid phase peptide synthesis (LPPS) approach offers a cost-effective process for large-scale manufacture by eliminating the need for specialized equipment and excess raw materials. Furthermore, this method permits standard analytical chemistry techniques that aren’t possible in SPPS, allowing robust in-process controls. Ultimately, choosing a liquid phase synthesis strategy can lower the overall cost of goods and facilitate tech transfer steps during the transition to commercial production.
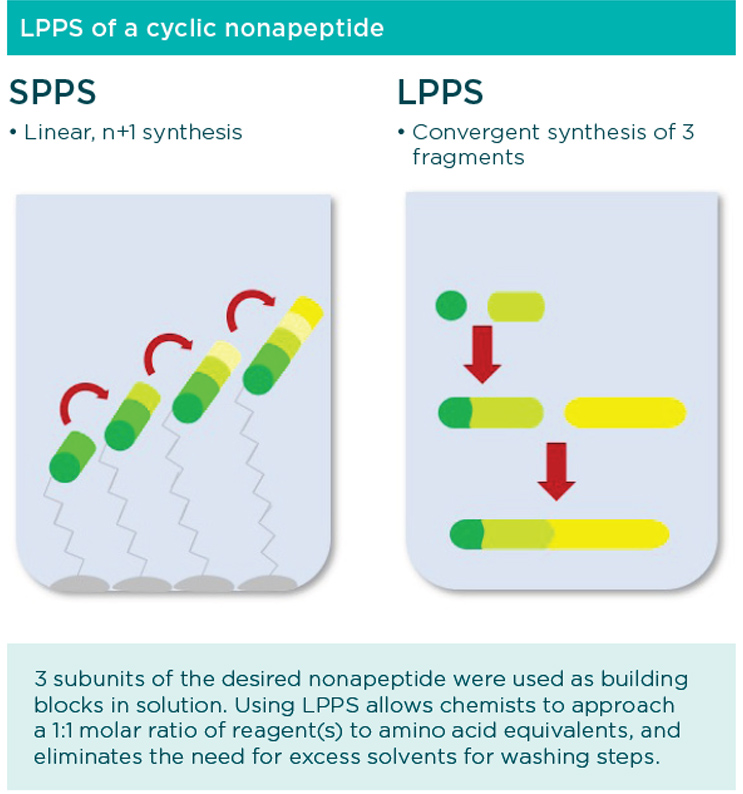
Linear vs convergent synthesis: Choosing the right synthesis strategy
Peptides in early stage development are often synthesized on a small scale using a solid phase strategy, adding amino acids one at a time while the growing peptide is attached to a supporting resin.
Solid phase synthesis requires specialized equipment to accommodate the steps of loading, coupling, cleavage, deprotection, purification and isolation. The attachment to the resin limits the ability to implement standard in-process controls during the reaction. Excess reagents and starting materials are required to achieve effective conversions in the synthetic transformations, and large volumes of solvent are necessary to rinse the resin efficiently between the coupling and deprotection steps. In addition, this method of synthesis requires a purification step (typically HPLC) and lyophilization. All of these requirements translate into exorbitant costs to synthesize the first kilogram of material, and could be cost prohibitive when considering scale-up for commercial production, or even for Phase 2 or Phase 3 studies.
By contrast, performing peptide synthesis in solution does not require specialized equipment. It can be performed in a normal GMP suite, easing the tech transfer process during later development. Instead of stringing together one amino acid at a time, this convergent strategy pools the necessary building blocks to create the desired API. Chemists can perform in-process controls using standard analytical chemistry techniques since the peptide is not attached to a polymer. Excess reagents are not required, in part because the iterative coupling, cleavage and deprotection steps are eliminated, which yields a significant cost advantage. Individual amino acids and intermediates can be competitively sourced from suppliers in multi-kilogram quantities. Scalable crystallization techniques replace the HPLC step to purify the end product, further increasing the cost efficiency.
When drug manufacturers are planning for late stage studies or commercialization, cost and scalability are critical factors in steering long-term strategies. The ability to source materials competitively while facilitating a scalable tech transfer process addresses the ultimate goal of lowering the cost of goods to achieve multi-kilogram or multi-ton production.
Successful delivery: Replacing SPPS with LPPS to supply bulk peptides
At Cambrex, our expert chemists design custom strategies to leverage the latest innovations in peptide synthesis for achieving high quality large-scale production at the lowest cost. By utilizing normal GMP suites during LPPS, methods are aligned with the tech transfer process and support preparedness for the eventual transition to commercial production. In this case, a convergent LPPS strategy was used to synthesize a 9-amino acid peptide from 3 fragments which included a 4-mer, a 3-mer, and a dipeptide tail. The advanced intermediates were combined in a few short steps to create the desired API.
When Cambrex adopted the project, the existing method to synthesize off the resin resulted in 60% purity and required HPLC with high quantities of solvents. Using the LPPS strategy, process development studies ensured that 99.5% purity was reproducibly achieved, and the chemists were able to avoid any HPLC or normal phase chromatographic purification steps. Instead, purification was performed using scalable crystallization methods by leveraging expertise in Cambrex’s Scotland facility that houses specialized capabilities in crystallization of peptides derived from LPPS.
The synthesis design was further optimized by using a single late-stage protection group, which enabled a reduction in final steps to only 1 global deprotection step (versus 2 steps in solid-phase synthesis). Ultimately, Cambrex executed their LPPS strategy concept to deliver a decrease in cost of raw materials by allowing competitive sourcing of simplified building blocks, and by facilitating the eventual tech transfer process for further development.
Building capabilities: Supplying a diverse market
Peptide-based therapies are no longer relegated to replacement hormones derived from animal origin. More recently, peptide drug candidates are indicated across a wide variety of therapeutic applications ranging from cancer and neurodegenerative diseases to antimicrobial agents. The promise of this class of molecules lies in harnessing their natural activity as endogenous regulatory molecules to mediate key biologic functions. The inherent specificity and short half-life of peptides bypasses frequent obstacles encountered during development of other drug classes, mainly nonspecific binding to non-target molecules and accumulation in tissues.
With strategic implementation of LPPS methods, CDMOs are poised to overcome hurdles in manufacturing costs to help drug producers succeed in scaling up production of their peptide candidates. Cambrex designs synthesis strategies to deliver a decreased cost of goods through a tech transfer package that plans for longterm commercial scale production.
Access the peptide market. Biology cleared the first hurdles of specificity and toxicity.
Cambrex’s synthesis capabilities can help eliminate the cost of goods hurdle.
About Cambrex
Cambrex is a leading global contract development and manufacturing organization (CDMO) that provides comprehensive analytical and IND enabling services, as well as drug substance development and manufacturing across the entire drug lifecycle.
With over 40 years of experience and a team of 2,000 experts servicing global clients from North America and Europe, Cambrex is a trusted partner in branded and generic markets for API development and manufacturing. Cambrex offers a range of specialized drug substance technologies and capabilities, including continuous flow, controlled substances, solid-state science, material characterization, and highly potent APIs.
