Design Of Experiments Approach To Enabling Studies For Process Validation
By Matthew Schiesher, Ph.D., Research Fellow, Cambrex

Process validation is a key step in preparing for commercial active pharmaceutical ingredient (API) production, requiring a significant depth of knowledge and data related to the manufacturing process. Appropriate design, execution, and interpretation of lab studies enable the definition of a process knowledge map and justify the operational region to ensure the API is manufactured in a safe and consistent manner, meeting all quality and regulatory requirements. And while compliance is always a top priority for sponsors, so too is developing an efficient process that can help reduce the late-stage development costs often incurred during API production.
In a recent project, Cambrex applied process improvement methodology to the process validation step in order to reduce costs for their client. The multi-faceted approach outlined below defined manufacturing parameters without the expense of surplus experimentation, as well as identified impactful process improvements that ultimately provided not only an optimized, robust process but also an economic advantage.
Data Interpretation Using Statistical Methodology
The increasing need for improved speed and efficiency in drug development has led to the adoption of various knowledge-driven, risk-based approaches in pharmaceutical manufacturing, such as the use of statistical methodology in API production. Not only can its application help develop the required data for regulatory authorities, but statistical methodology can also help manage the number of experiments, leading to reduced timelines and cost. Recognizing this, Cambrex employed a systematic approach in the following case study to develop the process knowledge map that would help prepare a multi-step API program for process validation. A standard evaluation of the results suggested the need for an additional series of studies. However, instead of relying solely on statistical interpretation, a pragmatic review of the data was undertaken from a process chemistry and chemical engineering perspective to understand how the information would impact process implementation in a manufacturing environment.
The blended, multi-disciplinary approach to data interpretation identified robust operating parameters suitable for validation at commercial scale without unnecessary time or cost of further experimentation. The process withstood a wider than anticipated range of operating conditions without impacting the quality of the API, reducing the risk of expense of investigation and additional purification during manufacturing.
Initial Procedure And Key Studies
The first step of the process was treatment of starting material one (SM-1), which is a carboxylic acid with oxalyl chloride and dimethylformamide (DMF) at just over ambient temperature. This reaction mixture is a slurry that is stirred out until the reaction conversion to the acid chloride passes the limit of not more than 15% residual SM-1. In parallel, another reactor is charged with amine hydrochloride starting material two (SM-2) in a blend of 2-methyltetrahydrofuran (2-MeTHF), water, and sodium hydroxide. The acid chloride slurry is then transferred into the freebase mixture. After a five-hour stir time, another reaction conversion test is performed with a limit of no more than 5% SM-2. Once the second test passes, the batch is filtered, washed, and dried. It is worth noting that, in the procedure provided to Cambrex as a starting point, SM-1 was always in slight excess of oxalyl chloride, which is why reaction conversion 1 has a relatively high tolerable limit of 15% residual SM-1. Rigorous control over the relative stoichiometry between SM-1, SM-2, and oxalyl chloride is also required.
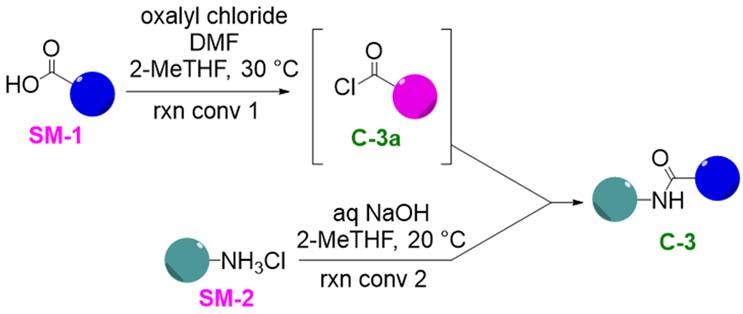
Figure 1: Step One Scheme
Table 1 outlines several key studies that were conducted to ready the process for a pilot plant batch, with the final lab trial (Trial 5) producing an R&D demonstration batch.
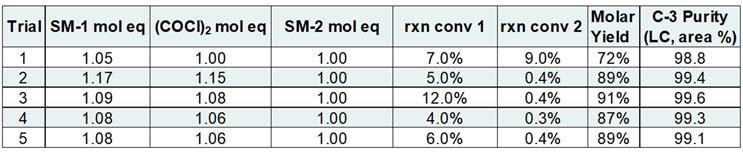
Table 1: Initial Process Evaluation Summary
The first process transfer experiment had successful reaction conversion 1 to the acid chloride, as well as good isolated yield and purity of intermediate C-3. Reaction conversion 2 (noted as rxn conv 2 in Table 1) did not meet specification. Trials 2 through 5 focused on adjusting the relative amounts of SM-1 and oxalyl chloride to realize a higher conversion of SM-2 to C-3. The goal was achieved, with less than 1% residual SM-2 becoming the typical reaction conversion 2 result. The updated conditions also improved upon molar yield and purity of C-3. Trial 5, conducted at 20-liter scale, was the final demonstration batch prior to running the process in the pilot plant.
Early in the lab transfer trials, the project team identified agitation as an important parameter to investigate for successful scale-up. Consistent with Schotten Baumann reactions, the mixture of 2-MeTHF, water, sodium hydroxide and SM-2 is fully soluble through biphasic. However, the conversion of SM1 to C-3a and C-3a to C-3 are both slurry-to-slurry reactions that require effective mixing. Therefore, data from the R&D demonstration batch were used to model the power-to-volume ratio for establishing the agitation settings in the production reactors. Once those calculations were completed, it was noted that the sheer rates and the micromixing times in the production equipment were comparable to those in the final lab demonstration trial. Based on the agitation study, the team concluded that mixing would not be an issue during scale-up.
Table 2 provides a comparison between the R&D demonstration and pilot batches. The R&D demonstration batch was executed on 0.9 kilograms of SM-2 while the pilot batch was executed on 28.8 kilograms (of SM-2).
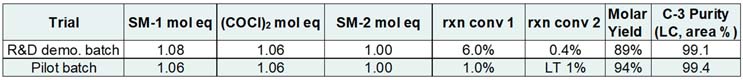
Table 2: Comparison between R&D demonstration batch and pilot batch
At pilot scale, reaction conversion 1 (noted as rxn conv 1 in Table 2) shows slightly better performance compared to the final lab trial. For reaction conversion 2, these numbers are essentially the same, with the difference in how sub-1% conversion is reported outside the R&D lab. After the successful pilot batch execution, the R&D focus switched to validation-readiness activities.
Process Validation Preparation
Spike, fate, and purge studies were the first step in preparing for process validation, where the goal was to track impurities in order to provide purge factors. The data would drive updates to specifications for starting materials, intermediates, and in-process controls. Several impurities were identified for further study:
- SM-1 had one impurity, SM-1-I1. The team did not believe this impurity would survive the reaction conditions but instead convert to SM-1-I1b, which was a corresponding impurity from SM-1-I1 reacting with oxalyl chloride.
- SM-2 was a bit more complicated with two impurities, SM-2-I1 and SM-2-I2. These were also expected to react in the process and each generate a new impurity, i.e., SM-2-I1b and SM-2-I2b.
- SM-2 had two additional impurities, SM-2-I3 and SM-2-I4, which were not expected to undergo any further conversion when exposed to the reaction conditions and should be monitored as-is.
- The final consideration was the consequence of excess of limiting reagent SM-2, as it was known to be tolerable up to a limit of only 5% (compared to a 15% limit for SM-1).
The entire study was completed in only three trials (results outlined in Table 3). At the time, there was uncertainty about whether impurity preparation and characterization should be done to determine response factors or if a specific wavelength should be used to calculate purge factors. The team ultimately decided that, instead of the effort to determine the relative response factor for each individual impurity, including in situ reaction-generated impurities, multiple UV wavelengths would be used when analyzing the samples isolated from the spike fate and purge trials. Taking a conservative approach, data from the worst-case wavelength were used for calculating each purge factor.
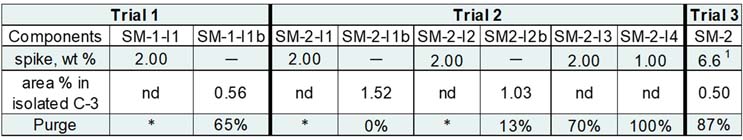
Table 3: Spike, Fate, and Purge Trial Summary
In Trial 1, the team evaluated the impact of spiking additional 2% by weight of impurity SM-1-I1 prior to introducing oxalyl chloride. This impurity was consumed during the reaction, as expected, and not observed in the isolated C-3. Impurity SM-1-I1b was observed in the isolated intermediate at around 0.56%, leading to a purge factor of approximately 65% at the most conservative analytical wavelength. For Trial 2, impurities SM-2-I1, SM-2-I2, and SM-2- I3 were spiked into the batch at 2%; SM-2-I4 was added at 1%. SM-2-I1 and SM-2-I2 were fully consumed in the reaction and generated their analogous reaction product impurities, i.e., SM-2-I1b and SM-2-I2b. Again, purge factors were calculated at the most conservative wavelength, with final results of 0% and 13% respectively. Impurity SM-2-I4 had a 100% purge value whereas SM-2-I3 was at 70%.
Trial 3 required an excess of SM-2 going into the workup, which was spiked into the batch after reaction IPC 2 had passed. Only half of a percentage point was observed from the trial once C-3 was isolated, for a final purge value of 87%.
This purge data guided specification changes for SM-1, SM-2, and C-3 (no changes were made to the IPC limits at this time) ahead of validation. The next focus of the R&D studies was process mapping.
Process Mapping: Design of Experiments Studies
Cambrex’s general approach to process mapping is driven primarily by design of experiments (DoE). The goal of the screening phase in a DoE program is to identify the significant factors that are influencing the change in responses as variables are altered systematically.
Process mapping begins with a risk assessment, which establishes the responses, factors, and factor ranges that the team would like to evaluate. The responses are generally the IPC and isolated intermediate specification release tests. The factors to be studied are the different experimental variables. Together, the factors and responses define the scope of a screening study. Depending on the results of the screening, the program may then move to an optimization phase. In some cases, this follow-up study is needed to help establish a full multivariable design space.
The last phase, which is also conducted only as needed, is verification or single-factor trials. These trials can be used to show the model generated during the optimization study is performing as expected or to look at different experimental variables that do not fit into a DoE study. For example, drying temperature is a common variable not included in a multivariate study, where a sample may be dried at three different temperatures to ensure there is no degradation observed in order to establish a process boundary.
Risk Assessment: Response And Factor Ranking
For the DoE risk assessment, a team is formed, generally made up of client representatives, process subject matter experts (SMEs), and a DoE SME. The team may also include analytical and quality control representatives as well as other chemists who are familiar with the development history of the procedure under evaluation. Critical responses derived from the in-process controls and C-3 specifications are identified and ranked on a scale of 1 to 5, with 5 being the most important. For this particular assessment, the responses being ranked were reaction conversion 1 with a limit of no more than 15% (ranked at 4.0), reaction conversion 2 with a limit of no more than 5% (ranked at 4.9), and the purity of C-3, which is the primary release criteria to move intermediate C-3 into the next step (ranked at 4.9).
Once the response rankings were established, the team identified the various factors that could influence these three responses. The selected factors were then also ranked 1 to 5, indicating their likelihood of impacting each of the responses. The response and factor rankings used to generate the weighted averages are shown in Table 4 and help define the scope of the screening study.
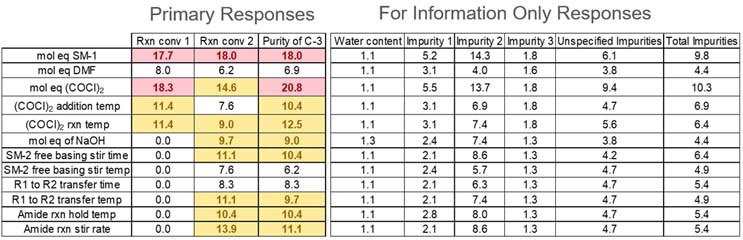
Table 4: Weighted Averages for Primary and FIO Responses
Weighted average values with a white background are those less than 9 and do not need to be included in the screening study; yellow indicates values between 9.0 and 14.9 and should be considered for the screening study by the project team; values with a red background are those 15.0+ and should be included in the screening study.
Note: Responses on the right portion of Table 4 are “for information only” and are not considered in decisions of scope or design of the DoE study.
For those risk factors in yellow, otherwise known as “medium” risk factors, a systematic approach is employed to evaluate whether they are to be included in the screening study. Information, such as supplemental studies or historical process data, is often cited to justify the decision. It is not recommended to arbitrarily include all medium risk factors, as this increases the number of trials. The decision-making process is documented to assist in answering any questions posed by regulatory agencies regarding the factor selection procedure.
Following protocol, the team considered all medium risk factors for inclusion in the screening DoE Study. In this case, it was decided that the oxalyl chloride addition temperature would be combined with the reaction temperature and not studied separately. Since the reaction temperature was going to be included, the team would use that factor’s result as a guide for how to control the Tmax during addition as it was already known to be readily dose controlled. The process, which was known to work well when high and low amounts of sodium hydroxide are charged, was not included in the screening study. The SM-2 free basing stir time was subjected to a supplemental study prior to finalizing the scope of the screening trials and was found to have no effect on the downstream processing between 0.5 and 18 hours. The team opted not to include the free basing stir time. The amide reaction stir rate was also investigated previously, and the mixing models were effective in transferring the process from the 20-liter chemical development demonstration batch into the pilot plant reactors. Repeating the amide reaction stir rate investigation was deemed unnecessary. Amide reaction hold time was also excluded from the study due to historical data that indicated the additional IPC samples can be run for up to 19 hours without detriment. Finally, it was determined that temperature transferring the oxalyl chloride reaction mixture into the amide coupling freebase would be included in the screening study.
Screening DoE: Experimental Design
Table 5 shows the factors selected, the current set points, and the factor ranges chosen for the screening DoE study.
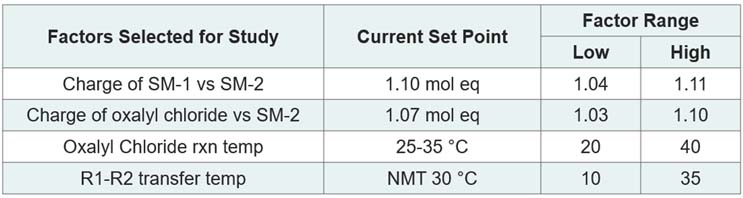
Table 5: Screening Study Factors and Ranges
Various approaches may be used to select the factor ranges, such as a general three or Six-Sigma-style approach. One may also use historical process knowledge. As a CDMO, Cambrex prefers to work with its customers to understand what they would like to study in the context of existing process knowledge. A wider factor range is generally advantageous for screening studies; however, in this type of study, it is important to not change the fundamentals of the chemistry. For example, one would want to avoid taking a solvent volume charge so low that the batch moves from a homogeneous solution to a heterogeneous mixture.
Table 6 shows the final results of the four-factor screening study in order of experiment execution.
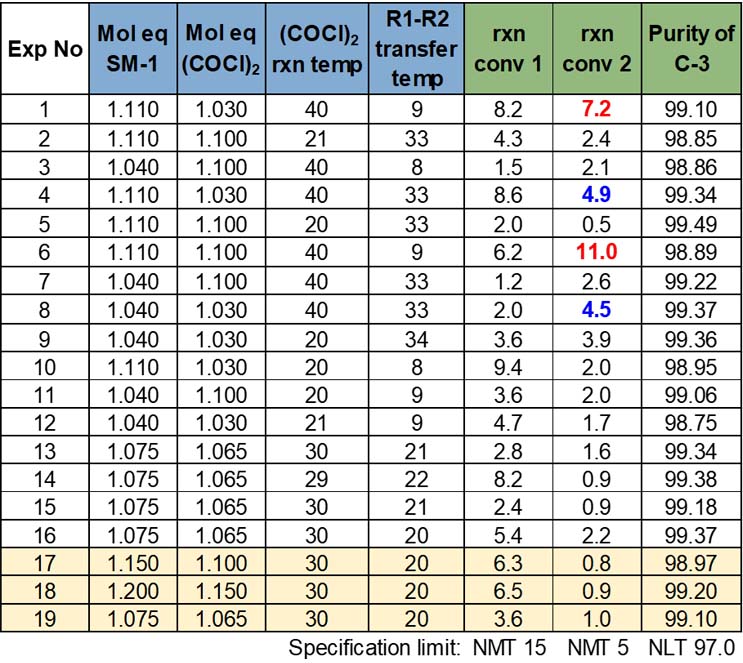
Table 6: Screening Study Results
A linear Plackett-Burman model was used for the screening study, which included 12 design runs and four center points (Trials 13 through 16). Cambrex uses a minimum of three center points; if there is more than one chemist executing the trials, two center points per chemist are used instead, as this allows the team to look at variability between individuals and the overall study. This model had a design power of 88, which exceeded the FDA requirement of 80. Some screening model options with fewer experimental runs were not considered as they did not meet the power criterion.
During a preliminary data review, there was some concern about reaction conversion 2, with two trials (displayed in red) that did not meet the NMT 5% criteria. Two additional trials were flagged (in blue) as near failures. With these data in mind, the project team decided to complement the initial data with a couple of extra design runs to further explore the impacts of changing SM-1 and the oxalyl chloride mole ratios. In this complimentary set of runs (highlighted rows at the bottom of Table 6), Trial 19 was also added as an extra center point to make sure there was no unusual variability versus the initial block of experiments. The complimentary trials passed criteria for all responses.
Overall, the purity of isolated C-3 met specification by a wide margin and showed no significant variability across the trials. The lack of variability resulted in no model being developed, but this is not concerning with all passing results. For reaction conversion 1, sufficient variability was obtained in the results to generate a model, but with all passing results, further analysis was not necessary. However, the project team still had to determine what led to the reaction conversion 2 failures and borderline passes.
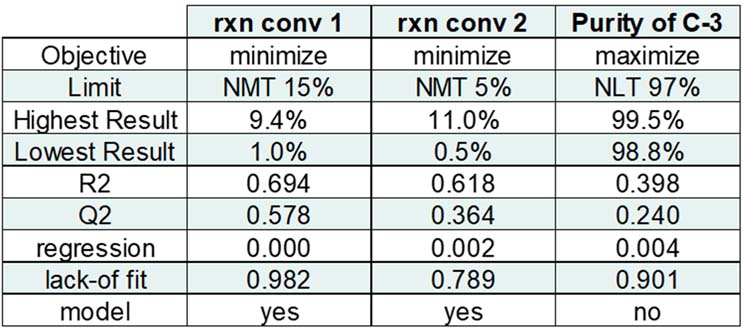
Table 7: Screening Model Data Summary
With a blend of passing and failing results, the team focused attention on the reaction conversion 2 data in order to define potential critical process parameters (CPPs) and proven acceptable ranges (PARs). During a review of the reaction conversion 2 model, three contributing model terms were found: oxalyl chloride mole equivalents, oxalyl chloride reaction temperature, and a square term. Common to many DoE screening models, the team learned that a square term influenced the results but not necessarily the square term’s identity. Further examination of the data showed that, while including oxalyl chloride mole equivalents provided a better model (higher R2 and Q2), it is not a significant model term. These reaction conversion 2 results were influenced by a single primary term, oxalyl chloride reaction temperature, and a square term. So, by process of elimination, the square term had to be oxalyl chloride reaction temperature, and that reaction temperature would have a non-linear influence on the results. With a single primary factor of significance remaining, the raw data were reviewed and re-sorted to better understand the oxalyl chloride reaction temperature impact on reaction conversion 2 (Table 8).
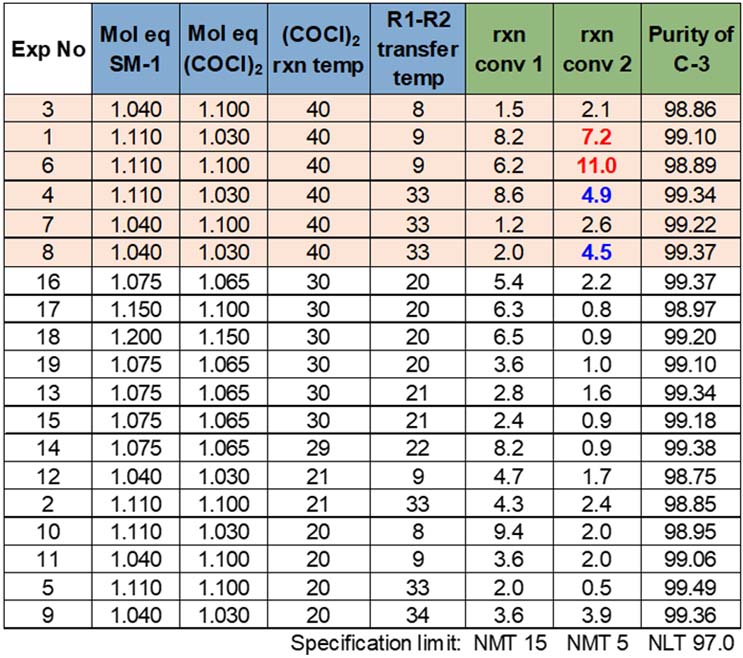
Table 8: Impact of Oxalyl Chloride Reaction Temperature
Of the 19 DoE experiments, six had an oxalyl chloride reaction temperature at 40o C. The two failures and two borderline cases were all found within those six trials. This data set indicates a 66% chance of passing the reaction conversion 2 at the high end of temperature range studied. All trials with the reaction temperature between 20 o C and 30 o C passed all three responses. This presented two potential options for moving forward. The first was to run additional experiments to quantify the impact of the model square term and identify the risk of failing reaction conversion 2 when the oxalyl chloride reaction temperature is operating between 30 o C and 40 o C. The other path was to set the oxalyl chloride reaction temperature proven acceptable range (PAR) at 20 o C to 30 o C. For the other 3 factors, PAR would be the screening study’s high/low and the program could be complete for this step.
To determine how best to define the reaction conversion 2 PAR, the team looked at the pilot plant batch data, specifically the oxalyl chloride reaction temperature, and found that it was easily maintained under 30 o C. The oxalyl chloride charge transfer temperature was easily dose controlled and there were no excursion events once the charge was complete. Therefore, the team decided that the risk of temperature excursions during the acid chloride reaction stir-out would not change when going from pilot plan to validation equipment. At the Cambrex Charles City manufacturing facility, the preferred operating temperature range is +/- 5 o C with a requested minimum of +/- 3 o C. The study ranges for the oxalyl chloride reaction fall within the preferred boundaries, so the reaction temperature PAR was set to 20 o C to 3 o 0 C, and this factor was designated as a potential CPP.
Due to economic importance, yield was also evaluated, despite being for information only. A complex model was obtained with these data. Three primary factors were statistically significant along with two cross terms and a square term. There is an opportunity to use this type of FIO data to help define the best normal operating ranges within the established proven acceptable ranges.
Conclusion
In this scope of work, the team at Cambrex looked at technology transfer to efficiently move the client process into the facility and ready it for its first run at manufacturing scale. While many experiments go into readying a procedure for a plant campaign, the process chemistry itself was demonstrated in nine key experiments to define the pilot batch conditions, including a successful lab demonstration batch. A successful pilot batch was executed thereafter, where API purity and yield expectations were met. This led to follow-up lab studies to generate spike, fate, and purge data that helped refine specifications for the next phase of the project. These studies tracked nine different impurities that were addressed in three experiments. A process mapping study that included 21 experiments was conducted to define PARs and one potential CPP. Finally, PAR highs and lows were vetted by the production group to ensure they were achievable, completing the process mapping effort at the screening phase.
